The electric cable used on a ship is specifically designed for marine installations both on shore and on ships. They are made considering the tough marine condition. On top, there are further risks of hazards related to dense smoke produce if the cables catch fire.
Even a small spark (discharge due to static charge) is dangerous on board ship. Having different electrical equipment and machinery increases this risk if not maintained to an order. Utmost care should be taken on a ship to avoid any scenario of electrical fire and damage to life and crew or property.
Electrical safety on board vessels is a vast topic with various aspects and applications. Here we will learn how electric cables react to fire On-board ships and how to avoid them.
Modern Power Electric Cable comes in a variety of sizes, materials, and types, each particularly adopted to its uses.
Cable consist of three major components.
- Conductor.
- Insulation.
- Protective Jacket.
The construction material of Electric cable is determined by three main factors, namely:
- The current carrying capacity determines the cross-section and size of the conductor.
- Environmental factors, such as sunlight, water, temperature, chemical, and mechanical impact.
- Working voltage determines the thickness of the insulation.
All-in-all, the make-up of each individual cable varies according to application.
The electric cable used on-board the vessel is required to be :
- Flame retardant.
- Fire proof.
- Low halogen content therefore low smoke emission.
- Oil resistant.
Ampacity : It is the current that a cable can carry continuously without the conductor exceed 80°C with an ambient air temperature of 45°C (i.e 35°C rise)
The recommended materials for Marine electrical cables on ships are
(a) Cable insulation
(i) Polyvinyl chloride
(ii) Ethylene polypropylene rubber
(iii) Cross-linked polyethylene
(iv) Low smoke, halogen-free cross-linked polypropylene rubber
(v) Silicon rubber
(vi) Cross-linked polyolefin
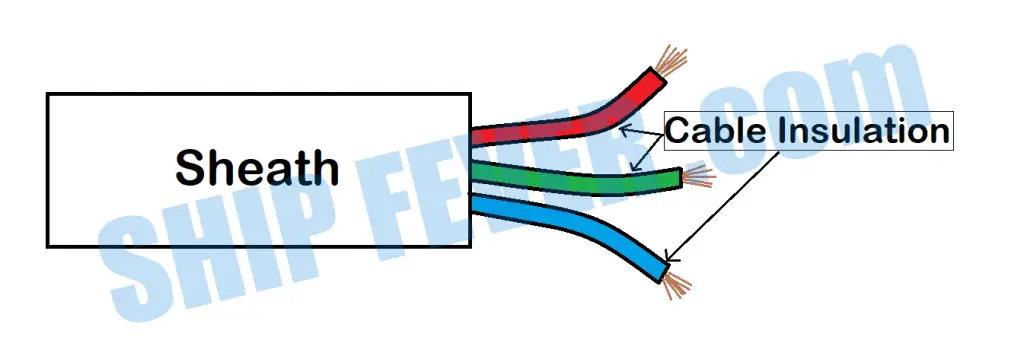
(b) Sheathing
(i) Polyvinyl chloride
(ii) Neoprene
(iii) Chloro-sulfonated polyethylene
(iv) Chlorinated polyethylene
(v) Cross-linked polyolefin
(vi) Polyolefin
Insulation: Insulation refers to the material wrapped around the individual wire. the purpose of insulation is to prevent the individual wire from touching each other.
Sheathing: Sheath refers to the material that wrapped around all the insulation and prevents it from any physical damage. The sheathing is what you can sea and touch.
A polymer undergoes combustion when there is sufficient energy in the form of heat to facilitate either decomposition or pyrolysis.
Decomposition or pyrolysis results in the release of volatile organic compounds that can subsequently be ignited.
The flammable products that evolve from the polymer during heating, mix with oxygen in the air and when their flammability limit is reached, they ignite in the presence of a flame or spark.
Once ignition has taken place, the burning process is exothermic. If sufficient energy is produced, it will overcome the endothermic requirements for polymer decomposition / pyrolysis and flame will be sustained and flame spread will be initiated
The following factors are of concern regarding reaction of electric cable to fire
► Heat release
► Flame spread
► Smoke production
► Acid gas production
► Toxic gas production

Heat Release Rate (HRR) – How much heat is released and how quickly is the single most important parameter which defines the hazard of a material. Some of the heat released during combustion is radiated back on to the fuel and directly affects fire growth.
Flame spread – Flame spread occurs when there is sufficient heat to sustain the polymer decomposition/pyrolysis process. Alternatively, the flame spread is affected by the heat of the combustion of the polymer. The heat of combustion represents the total energy that is released during the complete combustion of the material
Smoke production – Smoke is the result of incomplete combustion of polymer fuel. The flammability of a polymer or its ability to produce toxic fumes is determined by its chemical structure.
Ideally, aliphatic polymers that contain chains of oxygen have this tendency to produce low or less toxic fumes. On the other hand, polymers containing double-bonds or aromatic groups decompose, and release a large number of toxic fumes.

Smoke reduces visibility and hampers firefighters and can increase escape time for persons in burning space. Increasing the time required for escape increases the hazard from combustion gases and heat. Combustion gases are predominantly carbon monoxide, carbon dioxide, hydrogen cyanide, nitrogen oxides, hydrogen chloride, and sulfur dioxide.
Acidic gas production – Acidic gas production is of concern because of the post-fire damage which can be caused by electrical components from chemical corrosion. Cable sheathing typically contains a halogen, such as Bromine, Iodine, chlorine which produces toxic and corrosive products like HCl, during and after the fire. Smoke and soot toxicity and their potential effect on other shipboard materials and electrical components, long after the fire cleanup is also of concern.
Toxic gas production – Halogen based flame retardants are widely used as combustion inhibitors and they are very effective in suppressing ignition. However, if these flame retardant materials are exposed to severe or prolonged fire, enough to cause them to burn, then corrosive and irritant halogen acids are produced together with high levels of smoke and carbon monoxide.
Hence is the requirement for cables of “zero” halogen content. Cable specification states that chlorine + bromine + iodine shall be < 0.2% and fluorine < 0.1%.
Pyrolysis
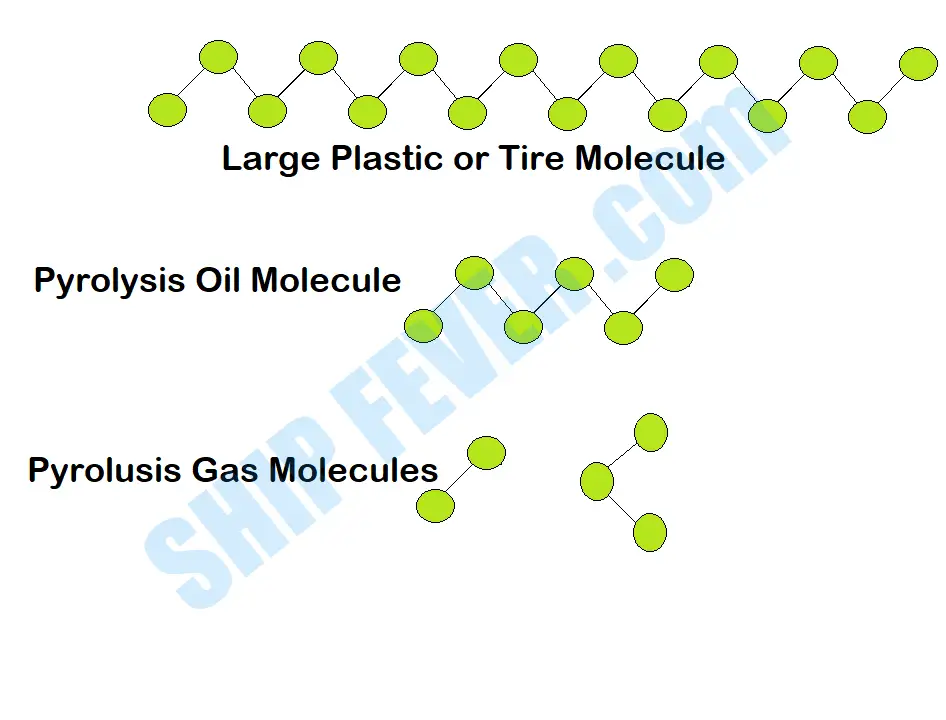 Pyrolysis is a chemical reaction that involves the breakdown of molecular structure of polymers from large molecule polymer structure to small molecular structure polymers with heat.
Pyrolysis is a chemical reaction that involves the breakdown of molecular structure of polymers from large molecule polymer structure to small molecular structure polymers with heat.
In a typical polymer structure, its molecules are in the vibrating stage at any given temperature. This is called molecular vibration that increase with the increase in temperature. The more the temperature the more the vibration of the molecules, therefore its frequency is directly proportional to temperature.
During pyrolysis, molecules are subjected to very high temperatures. This results in a subsequent increase in molecular vibration. At a time it reaches its peak vibration level beyond which it breaks and forms a smaller molecular chain.
Also Read:
- Best Outboard Motors | 7 Helpful Considerations
- What Is An Eductor And How Does It Work
- World Largest Container Ships | Top 10 List
- Types of Steam Turbine – A Basic Overview
- What Is Shipboard Oil Pollution Emergency Plan (SOPEP)

Elaborate classes of insulation…thanks!